Press-coated tablets are an advantageous technology to achieve delayed releases of active ingredients. They are characterized by a core–shell structure, that makes it possible to tune the lag-time and release kinetics in order to meet the chronotherapeutical goals.
Thus, these features are the most important quality attributes to be controlled when designing a press-coated tablet. Many studies have focused on the influence of the formulation on the release attributes.
At Skyepharma we have studied the influence of geometrical and process parameters on the release attributes of press-coated tablets, while keeping a constant formulation. In particular, the variation of compression pressure, layer thickness and band thickness made it possible to vary the lag-time from 1 h to 10 h.

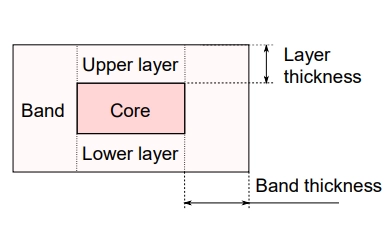
Process variables such as layer thickness, shell diameter, and compression pressure strongly influence both the lag time and the opening mechanism of press-coated tablets. Adjusting these parameters modifies the density distribution within the shell, which in turn governs whether the tablet opens via layer rupture or band opening. These two opening modes correspond to fast or slow release rates during dissolution testing.
The dissolution kinetics of the tablet core are directly dependent on the opening mode. Tablets exhibiting layer opening demonstrate a rapid release after the lag period, whereas tablets opening through a circumferential band release the drug more slowly. This relationship underscores the importance of visual observation during dissolution testing, as drug release data alone may not fully capture the underlying release mechanisms.
One of the advantages of press-coated tablets is the ability to modulate lag time without altering the core or shell formulations. By increasing compression pressure, layer thickness, or shell diameter, lag times ranging from 1 to 10 hours were successfully achieved. In addition to affecting the lag time, these parameters also influence the release kinetics after tablet opening, providing an additional tool for tailoring drug release profiles.
Given their significant influence on lag time and release behavior, geometrical parameters and compression pressure should be regarded as critical process parameters during development. Their careful optimization allows fine control over drug release attributes and reduces the need for reformulation, thereby streamlining development and scale-up.
At Skyepharma, we have several patented modified-release technologies that can be applied to your oral drug delivery platform. Our experts are motivated to study how we can apply our controlled release scientifique expertise as well as our innovation programs to optimize your oral drug delivery solution.