The MucoTabs Project: Targeting the GI Tract with Mucoadhesive Oral Tablets
Introduction of the study
Despite the widespread use of oral medicines, many drugs still face major challenges in achieving optimal absorption within the gastrointestinal tract.
Oral administration remains the preferred route for drug delivery due to its simplicity, non-invasive nature, and high patient acceptance. Among the different pharmaceutical dosage forms, tablets play a central role: stable, easy to store and transport, and relatively cost-effective, they represent the preferred option for a large majority of treatments.
However, not all active pharmaceutical ingredients fully benefit from this route of administration. Some molecules exhibit limited bioavailability, either because they are degraded in the stomach or because they are absorbed only in a specific region of the gastrointestinal tract. Others are sensitive to physiological variability, such as gastric pH or food intake, which can lead to incomplete or variable absorption.
It is in this context that one of our innovation projects was developed: MucoTabs, a research program carried out through an industry–academia collaboration between Skyepharma Production and LAGEPP, Université Claude Bernard Lyon 1.
The objective: to design mucoadhesive and pH-sensitive tablets capable of adhering to a specific region of the gastrointestinal tract and releasing the active ingredient in a prolonged manner. This approach combines scientific innovation, industrial feasibility, and strong clinical potential.
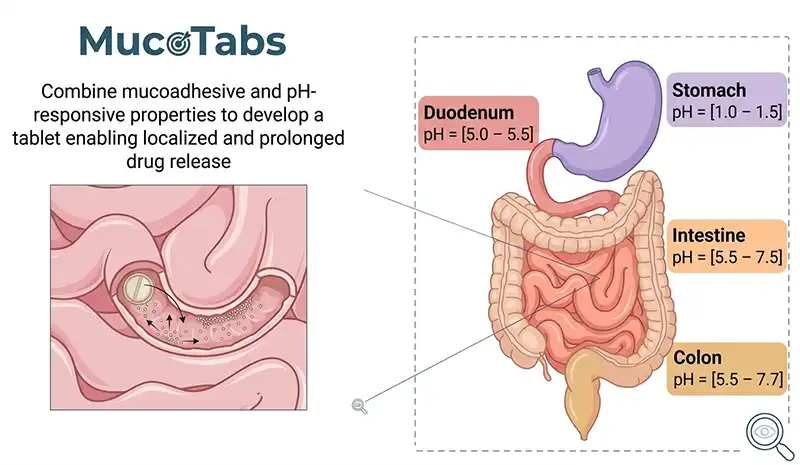
Combining mucoadhesion and pH sensitivity
Two complementary strategies were at the core of this project.
The first, mucoadhesion, enables the tablet to temporarily attach to the mucus layer lining the intestinal wall. This property increases the residence time in the targeted region and promotes controlled drug release.
The second strategy relies on pH sensitivity, exploiting the natural pH variations along the gastrointestinal tract to trigger the release of the active ingredient precisely where it is needed.
By combining these two mechanisms, we developed formulations capable of precisely targeting the stomach, intestine, or colon, maximizing therapeutic efficacy while reducing potential side effects.
Understanding mucoadhesion and pH sensitivity
Mucoadhesion refers to the ability of a dosage form to interact with the mucus layer that covers the gastrointestinal tract. Several mechanisms can explain this interaction: polymer hydration and swelling, adsorption of polymer chains onto the mucosal surface, diffusion and interpenetration into the mucus layer, electrostatic interactions, or mechanical anchoring within the microvilli of the mucosa.
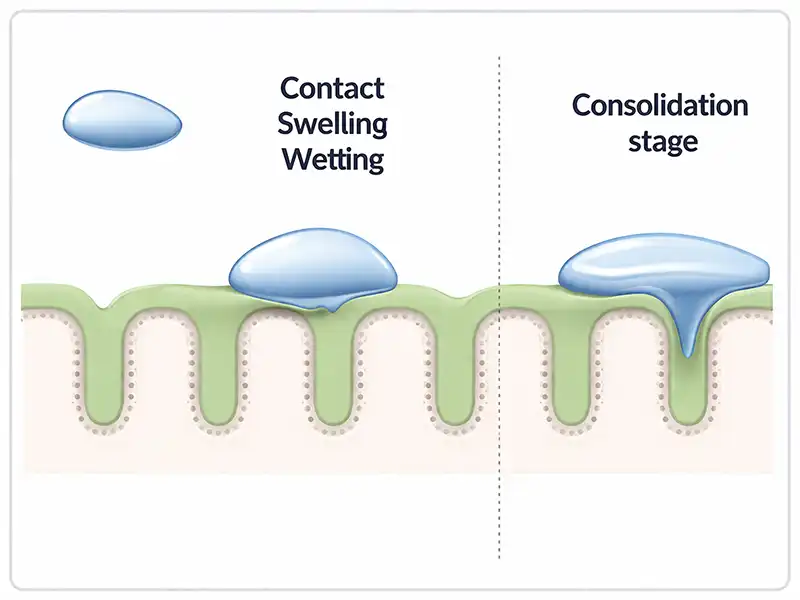
Together, these mechanisms allow the tablet to remain in place and release the drug in a controlled manner. The figure below illustrates the main mechanisms through which a mucoadhesive tablet can interact with mucus and mucosal tissues.
pH sensitivity takes advantage of the natural pH variations along the gastrointestinal tract. The stomach is highly acidic, while the small intestine and colon are more neutral or slightly alkaline. By selecting polymers that respond differently depending on the pH, tablets can protect the drug in unfavourable environments and release the active ingredient exactly where it is required, optimizing drug absorption.
From polymer selection to formulation optimization
The project began with a screening of commercially available polymers, analysing their physicochemical properties and their ability to interact with mucus.
The most promising polymers were then combined into binary and ternary mixtures to identify potential synergistic effects. A mixture design approach was used to optimize the proportions of the most effective polymer combination, while different diluents were tested to modulate hydration kinetics and enhance adhesion in the targeted region.
This pragmatic strategy ensures that the formulation can be rapidly translated to industrial scale.
Development of characterization methods and in vivo validation
Throughout the project, numerous in vitro and ex vivo characterization methods were developed and optimized to evaluate the performance of the formulated tablets.
These experiments allowed us to investigate key properties such as their adhesion to mucus, hydration kinetics, behaviour under simulated gastrointestinal conditions, and interactions with biological mucosal tissues.
These experimental approaches played a crucial role in understanding and optimizing the behaviour of the formulations, while enabling the selection of the most promising compositions.
The final validation step consisted of conducting in vivo studies in dogs, using tablets without active ingredients. These studies made it possible to observe the real behaviour of the tablets in the gastrointestinal tract and confirmed their ability to remain localized in the targeted region, demonstrating the potential of combining mucoadhesion and pH sensitivity for intestinal targeting.
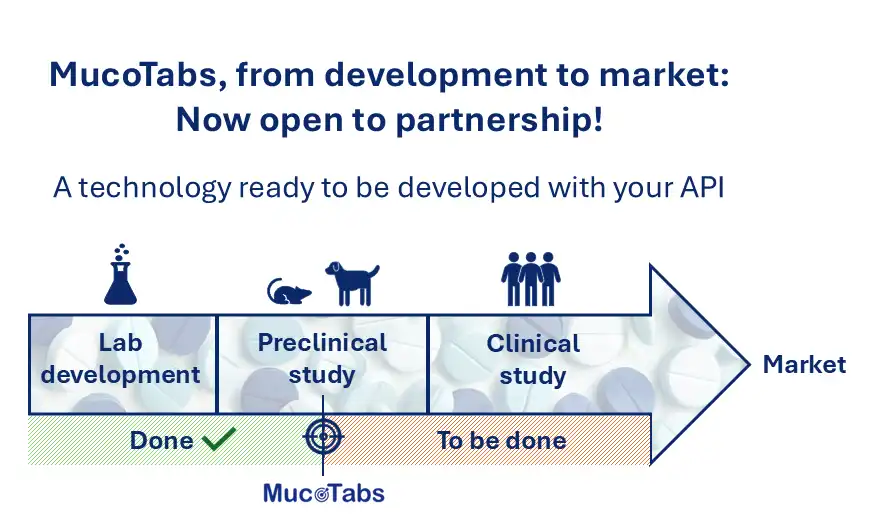
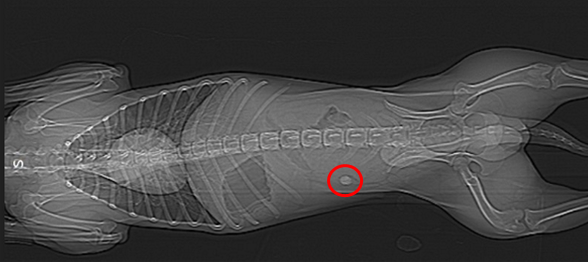
Concrete applications to improve patient care
Drugs with a narrow absorption window
Such as levodopa used in Parkinson’s disease: targeted release in the jejunum optimizes absorption and stability, potentially enabling nighttime dosing solutions where current treatments require patients to take a tablet every three hours.Several other drugs with narrow absorption windows could benefit from such technology, including furosemide (hypertension), acyclovir (herpes virus infection), metformin (diabetes), captopril (hypertension and heart failure), riboflavin (vitamin B2 deficiency) …
Local treatments for gastrointestinal diseases
- Inflammatory bowel diseases
- Colorectal cancer
- Helicobacter pylori infections
- Gastric ulcers
- …
In summary
The MucoTabs project demonstrated the feasibility of mucoadhesive and pH-sensitive tablets capable of precisely targeting regions of the gastrointestinal tract, optimizing drug release while remaining compatible with industrial manufacturing.
It opens the way toward more effective and better-tolerated treatments for a wide range of conditions, from inflammatory bowel diseases to drugs with narrow absorption windows.
By combining scientific rigor, technological innovation, and industrial vision, this work highlights how collaborative research can transform fundamental science into practical healthcare solutions.

